Deck 15: Dienes and Aromaticity
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/25
Play
Full screen (f)
Deck 15: Dienes and Aromaticity
1
Which compound should have the greatest max in its UV spectrum?
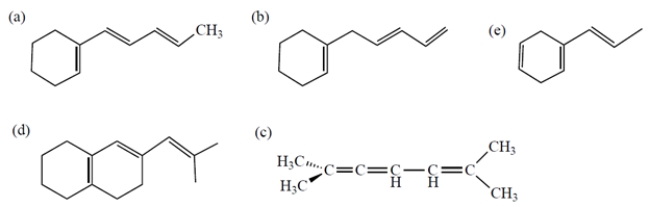
A) compound a
B) compound b
C) compound c
D) compound d
E) compound e

A) compound a
B) compound b
C) compound c
D) compound d
E) compound e
D
2
Which compound has UV absorption at the greatest wavelength?
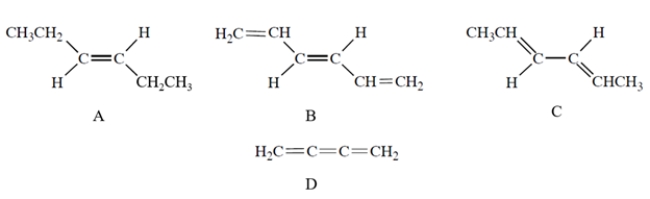
A) compound A
B) compound B
C) compound C
D) compound D

A) compound A
B) compound B
C) compound C
D) compound D
B
3
Circle the sp2-hybridized carbons in the structure.
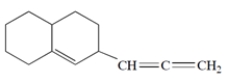

The central carbon of the cumulated double bond is sp-hybridized.
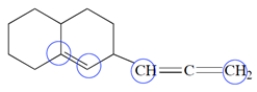

4
The diene reacts with HBr to give two isomers A and B (C6H11Br).
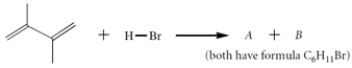 a. Give the structures of A and B.
a. Give the structures of A and B.
b. Give the structure of the carbocation intermediate involved in this reaction. Be sure to show any relevant resonance structures.
c. When compounds A and B are each treated under solvolysis conditions with acetone/water, each compound forms a mixture of the same two alcohols C and D. Give the structures of these two alcohols and explain why both are formed from each alkyl halide.
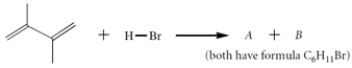 a. Give the structures of A and B.
a. Give the structures of A and B.b. Give the structure of the carbocation intermediate involved in this reaction. Be sure to show any relevant resonance structures.
c. When compounds A and B are each treated under solvolysis conditions with acetone/water, each compound forms a mixture of the same two alcohols C and D. Give the structures of these two alcohols and explain why both are formed from each alkyl halide.

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
5
Which compound or ion has five molecular orbitals?
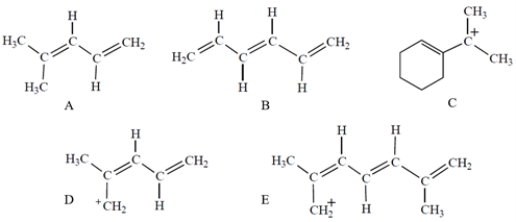
A) compound A
B) compound B
C) compound C
D) compound D

A) compound A
B) compound B
C) compound C
D) compound D

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
6
Which compound or ion has a UV/visible absorption at the greatest wavelength?
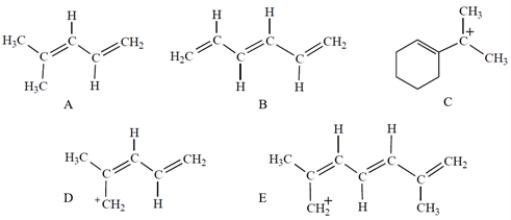
A) compound A
B) compound B
C) compound C
D) compound D
E) compound E

A) compound A
B) compound B
C) compound C
D) compound D
E) compound E

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
7
1,3-Butadiene, the simplest conjugated diene, has a UV absorption at 217 nm. (One nanometer = 10-9 meter.)
a. Calculate the energy of UV radiation with this wavelength (in kJ mol-1). Show your work.
b. Shown is an MO electron-occupancy diagram for the molecular orbitals of 1,3-butadiene. Draw an arrow on this diagram that corresponds to the energy of this UV radiation.
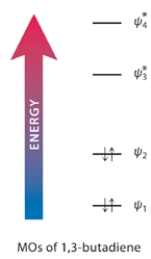
a. Calculate the energy of UV radiation with this wavelength (in kJ mol-1). Show your work.
b. Shown is an MO electron-occupancy diagram for the molecular orbitals of 1,3-butadiene. Draw an arrow on this diagram that corresponds to the energy of this UV radiation.


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
8
All but one of these molecules are active ingredients in commercial sunscreens. Which compound does not act as a sunscreen?
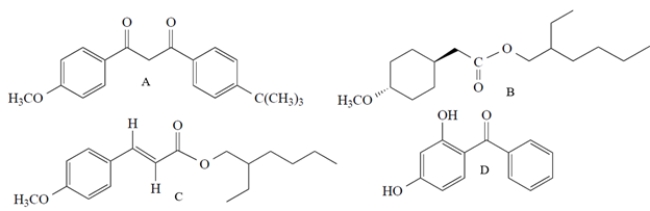
A) compound A
B) compound B
C) compound C
D) compound D

A) compound A
B) compound B
C) compound C
D) compound D

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
9
Which compound has the smaller (less positive or more negative) heat of formation?
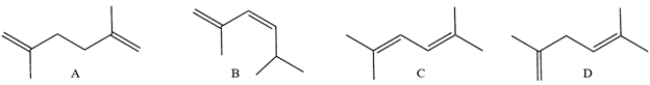
A) compound A
B) compound B
C) compound C
D) compound D

A) compound A
B) compound B
C) compound C
D) compound D

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
10
Select all structures that contain conjugated pi bonds.
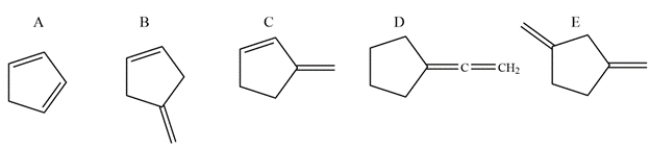
A) compound A
B) compound B
C) compound C
D) compound D
E) compound E

A) compound A
B) compound B
C) compound C
D) compound D
E) compound E

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
11
Draw the major organic product of this reaction.
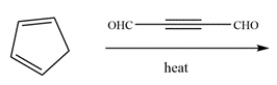


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
12
Give the structure of the diene and dienophile that would give this product after a Diels-Alder reaction.
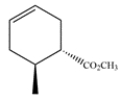


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
13
The Diels-Alder reaction can give two constitutional isomeric products, A and

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
14
Which of these dienes cannot undergo the Diels-Alder reaction.
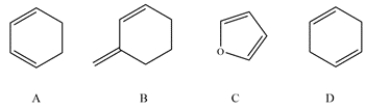
A) compound A
B) compound B
C) compound C
D) compound D

A) compound A
B) compound B
C) compound C
D) compound D

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
15
Draw the endo product formed in the Diels-Alder reaction. Clearly show stereochemistry, where applicable.
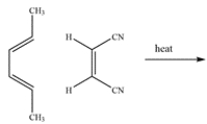


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
16
Deduce the starting materials that would give this compound via the Diels-Alder reaction.
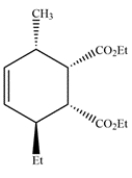


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
17
The reaction of an allylic alcohol with HBr generates the two products shown. Draw a curved arrow mechanism to explain how both products are formed.
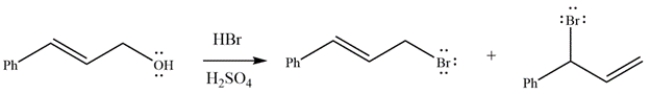


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
18
Draw the thermodynamic and kinetic product for the reaction. Clearly identify which is the thermodynamic and kinetic product.
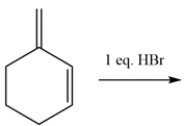


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
19
Which of these compounds is aromatic?
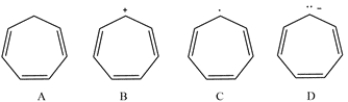
A) compound A
B) compound B
C) compound C
D) compound D

A) compound A
B) compound B
C) compound C
D) compound D

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
20
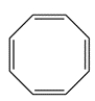
Cyclooctatetraene (COT) has eight pi electrons but is a stable compound. Explain why.


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
21
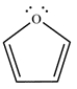
Draw a Frost circle and determine the molecular orbitals and electron occupancies for furan.


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
22
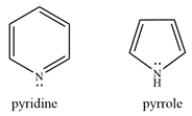
Pyridine and pyrrole are both aromatic nitrogen-containing rings. Explain how the lone pair on each nitrogen contributes or does not contribute to the pi electron count.


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
23
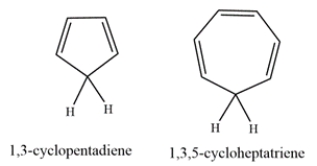
1,3-Cyclopentadiene is unusually acidic, with a pKa of ~16. On the other hand, 1,3,5-cyclohepatriene is much less acidic, with a pKa of ~36. Explain why the compounds have such differing acidities.


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
24
Determine whether each compound is aromatic, antiaromatic, or neither. Label each compound with the pi-electron count.
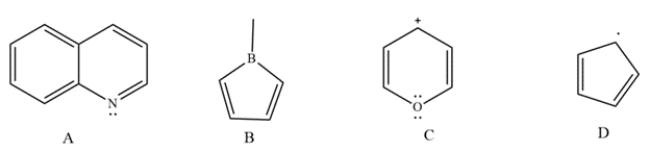


Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck
25
An arginine residue and a phenylalanine residue are located close to each other in a protein structure.
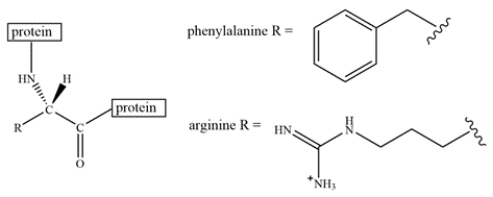 Select the most favorable interaction(s) of arginine and phenylalanine.
Select the most favorable interaction(s) of arginine and phenylalanine.
A) offset stacking
B) edge-to-face interaction
C) pi-cation interaction
 Select the most favorable interaction(s) of arginine and phenylalanine.
Select the most favorable interaction(s) of arginine and phenylalanine.A) offset stacking
B) edge-to-face interaction
C) pi-cation interaction

Unlock Deck
Unlock for access to all 25 flashcards in this deck.
Unlock Deck
k this deck


