Multiple Choice
The figure shows a pV diagram for 4.3 g of oxygen gas (O2) in a sealed container. The temperature T1 of the gas in state 1 is 21°C. What are the temperatures T3 and T4 of the gas in states 3 and 4? The ideal gas constant is R = 8.314 J/mol ∙ K = 0.0821 L ∙ atm/mol ∙ K, and the ATOMIC weight of oxygen is 16 g/mol. 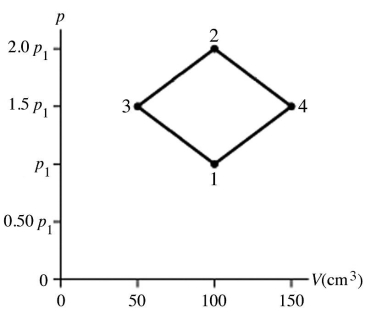
A) -52°C, 390°C
B) 16°C, 47°C
C) 220°C, 660°C
D) 11°C, 32°C
Correct Answer:

Verified
Correct Answer:
Verified
Q1: The hole for a bolt in a
Q7: The figure shows a 50-kg frictionless cylindrical
Q12: For a fixed amount of gas,if the
Q13: The coefficient of linear expansion of aluminum
Q16: A bag of potato chips contains 2.00
Q26: A 25-L container holds ideal hydrogen (H<sub>2</sub>)gas
Q30: If the temperature of an iron sphere
Q43: 3.00 moles of an ideal gas at
Q54: A machinist needs to remove a tight
Q57: A hot air balloon has a volume