Short Answer
The sulfide ion can react with the sulfur trioxide molecule to produce a thiosulfate ion, as shown below. 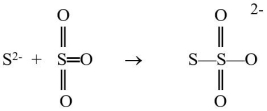 Which reactant is functioning as the Lewis base?
Which reactant is functioning as the Lewis base?
Correct Answer:

Verified
Correct Answer:
Verified
Q59: Which of the following would form a
Q60: What is the formula for the conjugate
Q61: What effect would bubbling SO<sub>3</sub> gas through
Q62: Which species would be expected to be
Q63: The conjugate base of HPO<sub>4</sub><sup>2-</sup> is<br>A)H<sub>2</sub>PO<sub>4</sub>.<br>B)H<sub>3</sub>PO<sub>4</sub>.<br>C)PO<sub>4</sub><sup>3</sup><sup>-</sup><sup>.</sup><br>D)PO<sub>4</sub><sup>2-</sup>.<br>E)H<sub>2</sub>PO<sub>4</sub><sup>-</sup>.
Q65: For the system HC<sub>6</sub>H<sub>5</sub>O + C<sub>4</sub>H<sub>7</sub>O<sub>2</sub><sup>-</sup> <img
Q66: Which species does not have a conjugate
Q67: Which solution should be the least acidic?<br>A)Al(NO<sub>3</sub>)<sub>3
Q68: For the system H<sub>3</sub>PO<sub>4</sub>(aq)+ COOH<sup>-</sup>(aq) <img src="https://d2lvgg3v3hfg70.cloudfront.net/TBW1039/.jpg"
Q69: Which species is amphoteric?<br>A)HNO<sub>3</sub>(aq)<br>B)Cl<sup>-</sup>(g)<br>C)CO<sub>3</sub><sup>2-</sup>(aq)<br>D)HSO<sub>3</sub><sup>-</sup>(aq)<br>E)PO<sub>4</sub><sup>3-</sup>(aq)