Multiple Choice
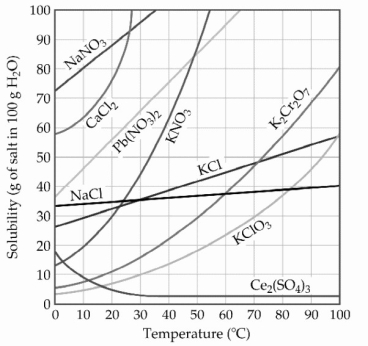
-The solubility of MnSO4 monohydrate in water at 20 °C is 70.0 g per 100.0 mL of water. A solution at 20 °C that is 4.22 M in MnSO4 monohydrate is best described as a(n) ________ solution. The formula weight of MnSO4 monohydrate is 168.97 g/mol.
A) hydrated
B) solvated
C) saturated
D) unsaturated
E) supersaturated
Correct Answer:

Verified
Correct Answer:
Verified
Q3: The dissolution of water in octane (C<sub>8</sub>H<sub>18</sub>)is
Q4: <img src="https://d2lvgg3v3hfg70.cloudfront.net/TB2701/.jpg" alt=" -The solubility of
Q6: Which component of air is the primary
Q10: A solution with a concentration higher than
Q11: A 1.35 m aqueous solution of compound
Q82: Which of the following choices has the
Q90: Of the following,a 0.1 M aqueous solution
Q110: Colligative properties of solutions include all of
Q156: A 0.100 m solution of which one
Q157: The principal reason for the extremely low