Essay
Consider the equilibria in CCl4, an apolar, aprotic, nondonor solvent.
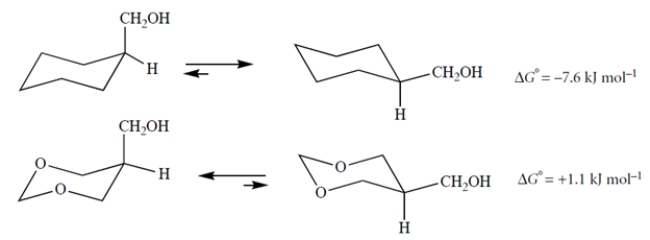 Explain why replacing the two carbons of the ring shown above with oxygens makes the axial conformation more stable.
Explain why replacing the two carbons of the ring shown above with oxygens makes the axial conformation more stable.
Correct Answer:

Verified
You might have noted that the oxygen lac...View Answer
Unlock this answer now
Get Access to more Verified Answers free of charge
Correct Answer:
Verified
View Answer
Unlock this answer now
Get Access to more Verified Answers free of charge
Q5: In which solvent should NaCl (an ionic
Q6: Sodium chloride, an ionic compound, should
Q7: Dissolving hexane in water has<br>A) a large
Q8: Draw the structure of 3-hexyn-2-ol.
Q9: Name the compound. Include the appropriate stereochemical
Q11: Draw all the alcohols having a molecular
Q12: When DMSO dissolves potassium chloride (K<sup>+</sup> Cl<sup>−</sup>),
Q13: Drugs are often conjugated to glucuronic acid
Q14: Name the compound with an IUPAC systematic
Q15: Arrange these compounds in order of increasing