Multiple Choice
Consider the reaction: N2O4(g) ? 2 NO2(g) 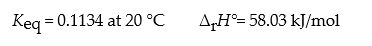
What is ?rG° for this reaction at 20 °C?
A) 0.36 kJ/mol
B) -5.4 kJ /mol
C) 4.9 kJ/mol
D) 5.3 kJ/mol
E) 5.4 kJ/mol
Correct Answer:

Verified
Correct Answer:
Verified
Q113: What is Δ<sub>r</sub>G°?<br><img src="https://d2lvgg3v3hfg70.cloudfront.net/TB5343/.jpg" alt="What is Δ<sub>r</sub>G°?
Q114: Which material has the largest entropy?<br>A)pure water<br>B)powdered
Q115: What is Δ<sub>r</sub>G° at 25 °C?<br>N<sub>2</sub>O<sub>4</sub>(g)→ 2
Q116: For CdO(s)+ SO<sub>3</sub>(g)→ CdSO<sub>4</sub>(s),Δ<sub>r</sub>H° = -279.4 kJ/mol,and
Q117: For CO(g)+ H<sub>2</sub>(g)→ H<sub>2</sub>CO(g),Δ<sub>r</sub>H° = -5.36 kJ/mol,and
Q118: In a sealed container,the rate of dissolving
Q119: Which of the following has the largest
Q120: Order the following by increasing entropy.<br>CO(g),COCl<sub>2</sub>(g),CO<sub>2</sub>(g),CaO(s)<br>A)CO<sub>2</sub> <
Q121: Consider the endothermic reaction:<br>N<sub>2</sub>(g)+ O<sub>2</sub>(g)→ 2 NO(g),Δ<sub>r</sub>H°
Q123: Choose the correct statements concerning entropy.<br>I.Following the