Multiple Choice
What is the mean free path for the molecules in an ideal gas when the pressure is 100 kPa and the temperature is 300 K given that the collision cross-section for the molecules of that gas is 2.0 × 10-20 m2? The Boltzmann constant is 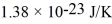 ,Avogadro's number is
,Avogadro's number is 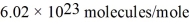 ,and the ideal gas constant is
,and the ideal gas constant is 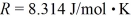 =
= 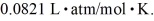
A) 1.1 × 10-6 m
B) 2.1 × 10-6 m
C) 1.7 × 10-6 m
D) 5.3 × 10-6 m
E) 1.5 × 10-6 m
Correct Answer:

Verified
Correct Answer:
Verified
Q1: At what temperature would the root-mean-square speed
Q2: If a certain sample of an ideal
Q3: An ideal gas is kept in a
Q4: An ideal gas is at a pressure
Q5: What is the total translational kinetic energy
Q7: An oxygen molecule falls in a vacuum.From
Q8: How many moles of water (H<sub>2</sub>O)molecules are
Q9: On a hot summer day,the temperature is
Q10: The interior of a refrigerator has a
Q11: Which contains more moles of material: 80