Exam 18: A Macroscopic Description of Matter
Exam 1: Concepts of Motion52 Questions
Exam 2: Kinematics in One Dimension59 Questions
Exam 3: Vectors and Coordinate Systems33 Questions
Exam 4: Kinematics in Two Dimensions50 Questions
Exam 5: Force and Motion30 Questions
Exam 6: Dynamics I: Motion Along a Line46 Questions
Exam 7: Newtons Third Law43 Questions
Exam 8: Dynamics Ii: Motion in a Plane20 Questions
Exam 9: Work and Kinetic Energy66 Questions
Exam 10: Interactions and Potential Energy55 Questions
Exam 11: Impulse and Momentum43 Questions
Exam 12: Rotation of a Rigid Body116 Questions
Exam 13: Newtons Theory of Gravity50 Questions
Exam 14: Fluids and Elasticity72 Questions
Exam 15: Oscillations49 Questions
Exam 16: Traveling Waves51 Questions
Exam 17: Superposition51 Questions
Exam 18: A Macroscopic Description of Matter46 Questions
Exam 19: Work, Heat, and the First Law of Thermodynamics96 Questions
Exam 20: The Micromacro Connection41 Questions
Exam 21: Heat Engines and Refrigerators44 Questions
Exam 22: Electric Charges and Forces26 Questions
Exam 23: The Electric Field32 Questions
Exam 24: Gausss Law41 Questions
Exam 25: The Electric Potential40 Questions
Exam 26: Potential and Field57 Questions
Exam 27: Current and Resistance32 Questions
Exam 28: Fundamentals of Circuits68 Questions
Exam 29: The Magnetic Field83 Questions
Exam 30: Electromagnetic Induction66 Questions
Exam 31: Electromagnetic Fields and Waves52 Questions
Exam 32: Ac Circuits44 Questions
Exam 33: Wave Optics51 Questions
Exam 34: Ray Optics60 Questions
Exam 35: Optical Instruments52 Questions
Exam 36: Relativity49 Questions
Exam 37: The Foundations of Modern Physics7 Questions
Exam 38: Quantization45 Questions
Exam 39: Wave Functions and Uncertainty18 Questions
Exam 40: One-Dimensional Quantum Mechanics32 Questions
Exam 41: Atomic Physics41 Questions
Exam 42: Nuclear Physics65 Questions
Select questions type
A fixed amount of ideal gas is held in a rigid container that expands negligibly when heated. At 20°C the gas pressure is p. If we add enough heat to increase the temperature from 20°C to 40°C, the pressure will be
Free
(Multiple Choice)
4.9/5  (33)
(33)
Correct Answer:
C
A glass flask has a volume of 500 mL at a temperature of 20°C. The flask contains 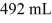 of mercury at
of mercury at 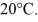 The temperature of the mercury and flask is raised until the mercury reaches the
The temperature of the mercury and flask is raised until the mercury reaches the 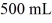 reference mark. The coefficients of volume expansion of mercury and glass are 18 × 10-5 K-1 and 2.0 × 10-5 K-1, respectively. The temperature at which this occurs is closest to
reference mark. The coefficients of volume expansion of mercury and glass are 18 × 10-5 K-1 and 2.0 × 10-5 K-1, respectively. The temperature at which this occurs is closest to
Free
(Multiple Choice)
4.9/5  (30)
(30)
Correct Answer:
A
You want to insert an aluminum rod, which at 20°C has a radius of 1.000200 cm into a copper tube which has a radius of 1.000100 cm at the same temperature. You decide to put both of them in the refrigerator. At what temperature will the rod just fit if both are cooled to the same temperature? The coefficient of thermal expansion for aluminum is 2.4 × 10-5 K-1, and that of copper is 1.7 × 10-5 K-1.
Free
(Multiple Choice)
4.8/5  (40)
(40)
Correct Answer:
E
Two steel spheres are made of the same material and have the same diameter, but one is solid and the other is hollow. If their temperature is increased by the same amount,
(Multiple Choice)
4.8/5  (39)
(39)
For a fixed amount of gas, if the absolute temperature of the gas is doubled, what happens to the pressure of the gas?
(Multiple Choice)
4.8/5  (25)
(25)
The figure shows a 50-kg frictionless cylindrical piston that floats on 0.68 mol of compressed air at 30°C. How far does the piston move if the temperature is increased to 300°C? 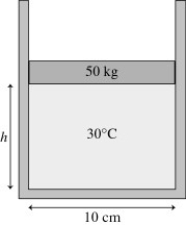
(Multiple Choice)
4.8/5  (36)
(36)
The coefficient of linear expansion of aluminum is 24 × 10-6 K-1 and the coefficient of volume expansion of olive oil is 0.68 × 10-3 K-1. A novice cook, in preparation for deep-frying some potatoes, fills a 1.00-L aluminum pot to the brim and heats the oil and the pot from an initial temperature of 15°C to 190°C. To his consternation some olive oil spills over the top. How much?
(Multiple Choice)
4.9/5  (39)
(39)
A 25-L container holds ideal hydrogen (H2) gas at a gauge pressure of 0.25 atm and a temperature of 0°C. What mass of hydrogen gas is in this container? The ATOMIC mass of hydrogen is 1.0 g/mol, the ideal gas constant is R = 8.314 J/mol ∙ K = 0.0821 L ∙ atm/mol ∙ K, and 1.00 atm = 101 kPa.
(Multiple Choice)
4.8/5  (33)
(33)
The figure shows a pV diagram for 4.3 g of oxygen gas (O2) in a sealed container. The temperature T1 of the gas in state 1 is 21°C. What are the temperatures T3 and T4 of the gas in states 3 and 4? The ideal gas constant is R = 8.314 J/mol ∙ K = 0.0821 L ∙ atm/mol ∙ K, and the ATOMIC weight of oxygen is 16 g/mol. 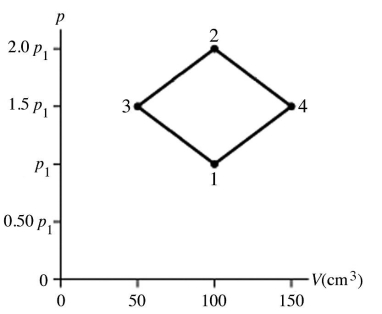
(Multiple Choice)
4.7/5  (37)
(37)
A machinist needs to remove a tight fitting pin of material A from a hole in a block made of material B. The machinist heats both the pin and the block to the same high temperature and removes the pin easily. What statement relates the coefficient of thermal expansion of material A to that of material B?
(Multiple Choice)
4.8/5  (29)
(29)
The hole for a bolt in a brass plate has a diameter of 1.200 cm at 20°C. What is the diameter of the hole when the plate is heated to 220°C? The coefficient of linear thermal expansion for brass is 19 × 10-6/°C. (Express your answer to 4 significant figures.)
(Multiple Choice)
4.8/5  (40)
(40)
A hot air balloon has a volume of 2.00 × 103 m3 when fully inflated, and the air inside the balloon is always at atmospheric pressure of 1.01 × 105 Pa because of the large opening used to fill the balloon and heat the air inside it. What is the mass of hot air inside the balloon if its temperature is 120°C? The universal gas constant is 8.314 J/mol ∙ K. (Assume a molecular weight of 28.8 g/mol for air.)
(Multiple Choice)
4.7/5  (36)
(36)
A bag of potato chips contains 2.00 L of air when it is sealed at sea level at a pressure of 1.00 atm and a temperature of 20.0°C. What will be the volume of the air in the bag if you take it with you, still sealed, to the mountains where the temperature is 7.00°C and atmospheric pressure is 70.0 kPa? Assume that the bag behaves like a balloon and that the air in the bag is in thermal equilibrium with the outside air. (1 atm = 1.01 × 105 Pa)
(Multiple Choice)
4.9/5  (28)
(28)
3.00 moles of an ideal gas at a pressure of 250 kPa are held in a container of volume of 25.0 L. The ideal gas constant is R = 8.314 J/mol ∙ K = 0.0821 L ∙ atm/mol ∙ K, and
1 atm = 1.01 × 105 Pa. The temperature of this gas is closest to
(Multiple Choice)
4.8/5  (35)
(35)
A 3.2-L volume of neon gas (Ne) is at a pressure of 3.3 atm and a temperature of 330 K. The atomic mass of neon is 20.2 g/mol, Avogadro's number is 6.022 × 1023 molecules/mol, and the ideal gas constant is R = 8.314 J/mol · K = 0.0821 L · atm/mol · K. The mass of the neon gas is closest to
(Multiple Choice)
4.7/5  (31)
(31)
Which contains more moles of material: 80 grams of helium gas (He, having atomic weight 4.0 g/mol) or 400 grams of argon gas (Ar, having atomic weight 40 g/mol)?
(Multiple Choice)
4.8/5  (41)
(41)
If a certain sample of an ideal gas has a temperature of 109°C and exerts a pressure of 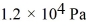 on the walls of its container, how many gas molecules are present in each cubic centimeter of volume? The ideal gas constant is 8.314 J/mol · K and Avogadro's number is
6.022 × 1023 molecules/mol.
on the walls of its container, how many gas molecules are present in each cubic centimeter of volume? The ideal gas constant is 8.314 J/mol · K and Avogadro's number is
6.022 × 1023 molecules/mol.
(Short Answer)
4.7/5  (32)
(32)
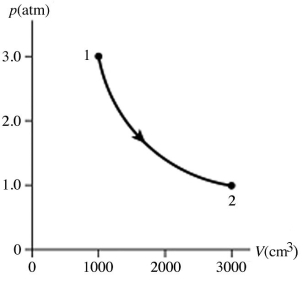
The figure shows a pV diagram for 0.95 mol of gas that undergoes the process 1 → 2. The gas then undergoes an isochoric heating from point 2 until the pressure is restored to the value it had at point 1. What is the final temperature of the gas? The ideal gas constant is R = 8.314 J/mol ∙ K = 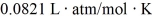 .
. 
(Multiple Choice)
4.9/5  (26)
(26)
Showing 1 - 20 of 46
Filters
- Essay(0)
- Multiple Choice(0)
- Short Answer(0)
- True False(0)
- Matching(0)