Exam 18: The First Law of Thermodynamics
Exam 1: Units, Physical Quantities, and Vectors107 Questions
Exam 2: Motion Along a Straight Line59 Questions
Exam 3: Motion in Two or Three Dimensions50 Questions
Exam 4: Newtons Laws of Motion44 Questions
Exam 5: Applying Newtons Laws95 Questions
Exam 6: Work and Kinetic Energy54 Questions
Exam 7: Potential Energy and Energy Conservation55 Questions
Exam 8: Momentum, Impulse, and Collisions50 Questions
Exam 9: Rotation of Rigid Bodies26 Questions
Exam 10: Equilibrium and Elasticity50 Questions
Exam 11: Fluid Mechanics50 Questions
Exam 12: Gravitation50 Questions
Exam 13: Periodic Motion50 Questions
Exam 14: Mechanical Waves44 Questions
Exam 15: Sound and Hearing66 Questions
Exam 16: Temperature and Heat63 Questions
Exam 17: Thermal Properties of Matter58 Questions
Exam 18: The First Law of Thermodynamics52 Questions
Exam 19: The Second Law of Thermodynamics50 Questions
Exam 20: Electric Charge and Electric Field58 Questions
Exam 21: Gausss Law41 Questions
Exam 22: Electric Potential55 Questions
Exam 23: Capacitance and Dielectrics52 Questions
Exam 24: Current, Resistance, and Electromotive Force50 Questions
Exam 25: Direct-Current Circuits53 Questions
Exam 26: Magnetic Field and Magnetic Forces36 Questions
Exam 27: Sources of Magnetic Field51 Questions
Exam 28: Electromagnetic Induction39 Questions
Exam 29: Inductance26 Questions
Exam 30: Alternating Current49 Questions
Exam 31: Electromagnetic Waves47 Questions
Exam 32: The Nature and Propagation of Light28 Questions
Exam 33: Geometric Optics81 Questions
Exam 34: Interference33 Questions
Exam 35: Diffraction49 Questions
Exam 36: Relativity51 Questions
Exam 37: Photons: Light Waves Behaving As Particles38 Questions
Exam 38: Particles Behaving As Waves52 Questions
Exam 39: Quantum Mechanics40 Questions
Exam 40: Atomic Structure41 Questions
Exam 41: Molecules and Condensed Matter31 Questions
Exam 42: Nuclear Physics89 Questions
Exam 43: Particle Physics and Cosmology44 Questions
Select questions type
The figure shows a pV diagram for 4.3 g of oxygen gas (O2) in a sealed container. The temperature T1 of the gas in state 1 is 21°C. What are the temperatures T3 and T4 of the gas in states 3 and 4? The ideal gas constant is R = 8.314 J/mol ∙ K = 0.0821 L ∙ atm/mol ∙ K, and the ATOMIC weight of oxygen is 16 g/mol. 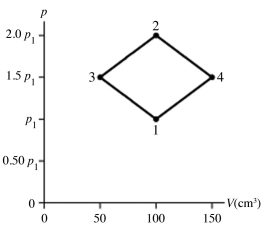
Free
(Multiple Choice)
4.8/5  (22)
(22)
Correct Answer:
A
A cylinder contains 24.0 moles of an ideal gas at a temperature of 300 K. The gas is compressed at constant pressure until the final volume equals 0.63 times the initial volume. The molar heat capacity at constant volume of the gas is 24.0 J/mol ∙ K and the ideal gas constant is R = 8.314 J/mol ∙ K. The change in the internal (thermal) energy of the gas is closest to
Free
(Multiple Choice)
4.7/5  (32)
(32)
Correct Answer:
A
When a fixed amount of ideal gas goes through an adiabatic expansion
Free
(Multiple Choice)
4.9/5  (33)
(33)
Correct Answer:
C
A quantity of ideal gas requires 800 kJ to raise the temperature of the gas by 10.0 K when the gas is maintained at constant volume. The same quantity of gas requires 900 kJ to raise the temperature of the gas by 10.0 K when the gas is maintained at constant pressure. What is the adiabatic constant γ for this gas?
(Multiple Choice)
4.9/5  (33)
(33)
An adiabatic compression is performed on an ideal gas. The final pressure is equal to 0.560 times the initial pressure and the final volume equals 1.50 times the initial volume. What is the adiabatic constant for the gas?
(Multiple Choice)
4.8/5  (36)
(36)
The figure shows a pV diagram for 0.0066 mol of gas that undergoes the process 1 → 2 → 3. What is the pressure p2. The ideal gas constant is R = 8.314 J/mol ∙ K = 0.0821 L ∙ atm/mol ∙ K. 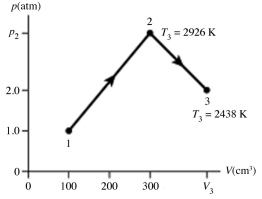
(Multiple Choice)
4.9/5  (38)
(38)
During an adiabatic process, 20 moles of a monatomic ideal gas undergo a temperature change from 450 K to 320 K starting from an initial pressure is 400 kPa. The ideal gas constant is R = 8.314 J/mol ∙ K.
(a) What is the final volume of the gas?
(b) How much heat does the gas exchange during this process?
(c) What is the change in the internal (thermal) energy of the gas during this process?
(Short Answer)
4.8/5  (30)
(30)
A cylinder contains 23 moles of an ideal gas at a temperature of 300 K. The gas is compressed at constant pressure until the final volume equals 0.43 times the initial volume. The molar heat capacity at constant volume of the gas is 24.0 J/mol ∙ K and the ideal gas constant is R = 8.314 J/mol ∙ K. The heat absorbed by the gas is closest to
(Multiple Choice)
4.9/5  (37)
(37)
During an adiabatic process, an ideal gas does 25 J of work. What is the change in the internal (thermal) energy of the gas during this process?
(Multiple Choice)
4.9/5  (34)
(34)
A container with rigid walls is filled with 4 mol of air with CV = 2.5R. How much does the internal (thermal) energy change if the temperature of the air rises from 16°C to 437°C? The ideal gas constant is R = 8.314 J/mol ∙ K.
(Multiple Choice)
4.8/5  (46)
(46)
An ideal gas with γ = 1.67 is initially at 0°C in a volume of 10.0 L at a pressure of 1.00 atm. It is then expanded adiabatically to a volume of 10.4 L. What is the final temperature of the gas?
(Multiple Choice)
5.0/5  (38)
(38)
A certain amount of ideal monatomic gas is maintained at constant volume as it is cooled from 455K to 405 K. This feat is accomplished by removing 400 J of heat from the gas. How much work is done by the gas during this process? The ideal gas constant is R = 8.314 J/mol ∙ K.
(Multiple Choice)
4.9/5  (35)
(35)
The figure (not to scale) shows a pV diagram for 1.8 g of helium gas (He) that undergoes the process 1 → 2 → 3. Find the value of V3. The ideal gas constant is R = 8.314 J/mol ∙ K = 0.0821 L ∙ atm/mol ∙ K, and the atomic weight of helium is 4.0 g/mol. 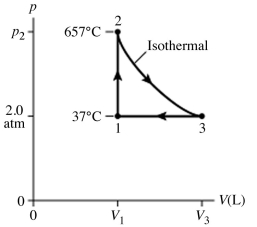
(Multiple Choice)
4.8/5  (43)
(43)
The figure shows the pV diagram for a certain thermodynamic process. In this process, 1500 J of heat flows into a system, and at the same time the system expands against a constant external pressure of 9.00 × 104 Pa. If the volume of the system increases from 0.020 m3 to 0.050 m3, calculate the change in internal (thermal) energy of the system. If the internal (thermal) energy change is nonzero, be sure to indicate whether this energy change is positive or negative. 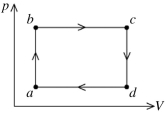
(Short Answer)
4.7/5  (45)
(45)
When an ideal gas increases in volume at constant pressure, the average kinetic energy of the gas molecules
(Multiple Choice)
4.9/5  (37)
(37)
A system has a heat source supplying heat to an ideal gas at a rate of 187.0 W and the gas is doing work at a rate of 130.9 W. At what rate is the internal (thermal) energy of the gas changing?
(Multiple Choice)
4.8/5  (33)
(33)
An ideal gas initially at 300 K and occupying a volume of 20 L is adiabatically compressed. If its final temperature is 400 K and γ = 1.30, what is its final volume?
(Multiple Choice)
4.8/5  (29)
(29)
An ideal gas is compressed in a well-insulated chamber using a well-insulated piston. This process is
(Multiple Choice)
4.8/5  (40)
(40)
In a thermodynamic process involving 7.8 moles of an ideal gas, the gas is at an initial temperature of 24°C and has an initial volume of 0.040 m3. The gas expands adiabatically to a volume of 0.080 m3. For this gas, CV = 12.27 J/mol · K, and the ideal gas constant is R = 8.314 J/mol ∙ K. Calculate the work done by the gas during this expansion.
(Short Answer)
4.9/5  (39)
(39)
A fixed amount of ideal gas goes through a process abc. In state a, the temperature of the gas is 152°C, its pressure is 1.25 atm, and it occupies a volume of 0.250 m3. It then undergoes an isothermal expansion to state b that doubles its volume, followed by an isobaric compression back to its original volume at state c. (Hint: First show this process on a pV diagram.) The ideal gas constant is 8.314 J/mol ∙ K, and 1.00 atm = 1.01 × 105 Pa.
(a) How many moles does this gas contain?
(b) What is the change in the internal energy of the gas between states a and b?
(c) What is the net work done on (or by) this gas during the entire process?
(d) What is the temperature of the gas in state c?
(Short Answer)
4.7/5  (35)
(35)
Showing 1 - 20 of 52
Filters
- Essay(0)
- Multiple Choice(0)
- Short Answer(0)
- True False(0)
- Matching(0)